Juveena® is an advanced therapy designed specifically for the prevention of the formation and reformation of intrauterine adhesions (IUAs).

ELEVATING UTERINE HEALTH
About the Juveena™ Hydrogel System
Prevent IUAs
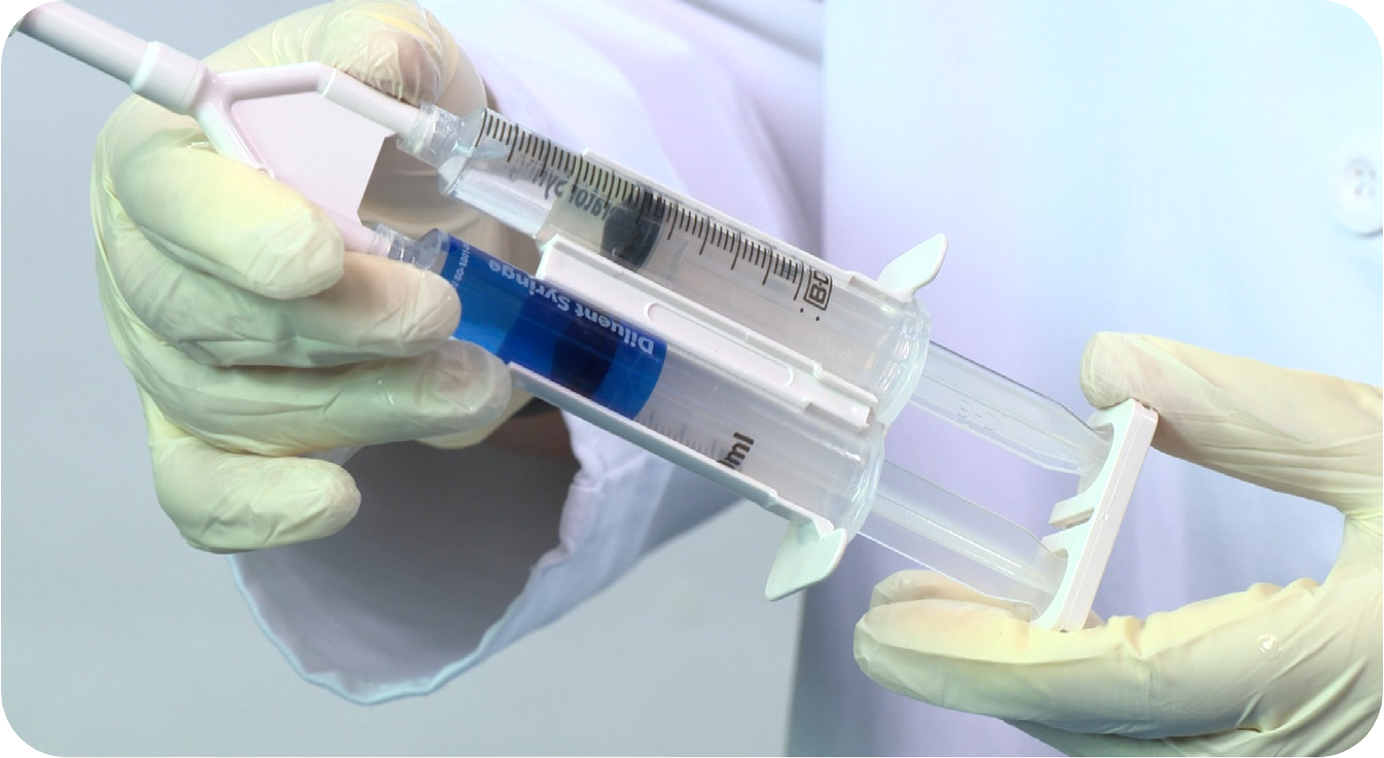
The Juveena Hydrogel System (HS), is intended for use as a temporary implant to prevent the formation or reformation of intrauterine adhesions (IUAs) following transcervical gynecological procedures.
Proprietary
The Juveena Hydrogel System consists of a proprietary transcervical catheter and two liquid precursors that fill the uterine cavity and immediately cross-link to form a soft temporary hydrogel implant that separates the uterine walls and prevent intrauterine adhesions from forming during the healing process.
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.
KEY BENEFITS / EFFECTIVE PREVENTION
Juveena is intended to prevent the formationof IUAs, addressing a critical concern for both physicians and their patient.

The low-profile, transcervical catheter ensures accurate placement and comfort for the patient

The hydrogel conforms to the uterine cavity and remains in place to create a favorable environment for the uterine lining to heal by providing both space and time for recovery.

The composition of the hydrogel is designed to liquify after 2-3 weeks (or one menstrual cycle) therefore no secondary follow-up procedure needed for removal.

Clinicians and women can trust using Juveena after transcervical procedures to reduce the risk of developing or IUAs. Juveena has been clinically evaluated in women with moderate to severe adhesions or who had fibroids removed.
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.
What are intrauterine adhesions(IUAs)?
Intrauterine Adhesions (IUAs), happen when scar tissue forms inside the uterus or cervix.1-3 This can occur after an injury to the uterus, such as surgery or a complicated pregnancy, which causes the lining to heal with bands of tissue. These adhesions can be thin or thick and may cover small or large areas. Doctors classify IUAs as mild, moderate, or severe based on how much scar tissue is present.
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.
IUAs often happen after procedureslike dilation and curettage (D&C),which may be done for:
Elective pregnancy termination
Treatment of a miscarriage
Removal of a retained placenta after delivery
IUAs can also develop after surgeryfor:
Fibroid removal
Excessive uterine bleeding3,4
Endometrial cancer testing
Uterine Septums
Repeat adhesion removal
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.
INTRAUTERINE ADHESIONS: CAUSES, RISKS, AND PREVALENCE
How Common Are IUAs?
IUAs often go unnoticed, especially in women who are not trying to conceive. Symptoms include lighter-than-normal periods (hypomenorrhea) or no periods at all. Standard exams and ultrasounds may not detect IUAs, leading to underdiagnosis.
IUAs and Pregnancy

after a late miscarriage

in women needing follow-up surgery after delivery or miscarriage

of women after a first-trimester pregnancy termination
IUAs and Pregnancy

after the second hysteroscopic resection

after the first hysteroscopic resection
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.
Who Is at Higher Risk?
Women with placental abnormalities, such as Placenta Accreta, are at greater risk because the placenta attaches more deeply to the uterus, making it harder to remove.4,5
Repeated procedures, including multiple D&Cs, also increase the likelihood of developing IUAs.
Higher risk with multiple surgeries
Placental abnormalities increase the chance of IUAs
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.
ABSTRACTS & PUBLICATIONS
Why Juveena
AAGL Podiums/Abstracts
UTERINE ADHESION RESOURCES
Patient and Physician Communities
The Juveena Hydrogel System in Pending FDA Approval. Not Available For Sale. Investigation Use Only.